April 25, 2017 — Ten infants in the neonatal intensive care unit of an Orange (CA) hospital have tested “positive” for a potentially deadly bacteria, the Los Angeles Times reported two weeks ago.
This newspaper’s report was the first time that the public learned of this MRSA outbreak in the NICU of the UC Irvine Medical Center.
Last week, I was interviewed by KNX 1070 CBS Radio (Los Angeles, CA), which asked me my perspectives about this just-disclosed outbreak.
During the interview, I raised a number of issues and questions for the public’s consideration. Whether KNX has posted a podcast or news article featuring my comments about this potentially deadly MRSA outbreak is unclear.
Briefly, ten (10) of the hospital’s infants were infected (or colonized) with the same MRSA strain, according to a Los Angeles Times article published two weeks ago on April 13th.
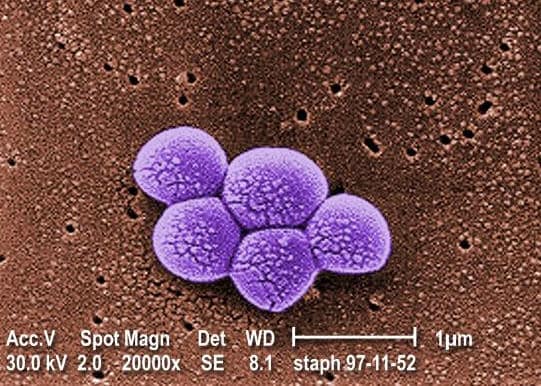
MRSA is an acronym for methicillin-resistant Staphylococcus aureus.
While already critically ill, all of the infants became infected with the outbreak’s MRSA strain while at the hospital, according to the Times.
Technical Advisor: Dr. Muscarella provides expert opinions, case reviews and advice for hospitals, device manufacturers and consumers about the causes of healthcare-associated infections (“HAIs”) linked to contaminated duodenoscopes, among other devices. Contact him (using your smart phone): Email | Text (SMS) | Skype
While this outbreak’s first MRSA case was identified last August according to the Orange County Register newspaper — with two neonates reportedly testing “positive” for the resistant bacterium as recently as last month — the Times reports that the public was not informed of this potentially lethal outbreak until two weeks ago, or eight months later.
In another related article, the Times reported that the hospital had advised its staff “to inform parents of the babies sickened or colonized by the bacteria about the ongoing outbreak.”
But the mother of one of the 10 affected infants disputes the hospital’s claim that parents were told about this MRSA outbreak, the Times also recently reported.
Moreover, according to the newspaper’s April 13th article, Orange County health officials said “they did not notify the public about the outbreak because they had no evidence that infants being treated at UCI’s neonatal unit were at higher risk than infants admitted elsewhere.”
To some, this rationale may be confusing.
Quality, Safety and Case Reviews: Click here to read about Dr. Muscarella’s quality and safety services design to help hospitals, manufacturers and the public reduce the risk of healthcare-associated infections, including superbug outbreaks linked to contaminated reusable medical equipment.
The mortality rate of MRSA infections can be as high as 30% according to the federal Centers for Disease Control and Prevention (CDC).
This rate notwithstanding, UC Irvine officials told the Times that none of the ten (10) neonates infected (or colonized) with the outbreak’s MRSA strain died.
Questions raised
The decision by public health officials not to publicly disclose potentially deadly bacterial outbreaks such as UC Irvine’s as reported by the Times, raises a number of questions, including:
- the merit of the informed consent process
- whether transparency in health care remains an important principle
- whether the public (at least in California) has implicitly granted hospitals and doctors (through passed legislation) the right at times to withhold clinical information that could be important for consumers to make educated and cost-effective decisions about their health care and treatment options.
The Times further reported that, according to state officials, UC Irvine’s handling of this 8-month long MRSA outbreak did not violate any federal or state laws.
Lack of infection data does not assure safety
According to the April 13th Los Angeles Times article, Orange County health officials did not notify the public about UC Irvine’s MRSA outbreak because these officials had “no evidence” that infants being treated in the hospital’s NICU were at a higher risk of contracting a MRSA infection (compared to the baseline infection risk).
This assessment raises a few points for discussion:
The lack of data or evidence confirming an infection risk does not assure the data are lacking and that patients are safe.
Moreover, a health department’s rationale for not informing the public, or the affected families, of a potentially lethal MRSA outbreak in a hospital’s NICU should be based, in my opinion — not on the absence of evidence showing a danger — but on the presence of scientific data confirming safety and that the hospital’s infants were indeed not at an increased risk of a MRSA infection.
To be clearer, a lack of data confirming an infection risk does not assure the patients are not at risk.
Based on the available information about this California hospital’s MRSA outbreak published to date, it would seem that the possibility cannot be summarily ruled out that infants being treated in the hospital’s NICU at the time of this outbreak, which began last August, were at an increased risk of a MRSA infection, the health officials comments notwithstanding.
The MRSA outbreak’s source
Whether health officials should have notified the public and affected families sooner about this hospital’s MRSA outbreak can be reasonably argued, if not because of at least one particular concern:
According to news reports, health officials still have not yet identified the source of UC Irvine’s MRSA outbreak, which began eight months ago.
As reported first by the Times, the hospital’s doctors “have not found the source of the infections – which continued even after 220 employees used antiseptic soap and ointment to eliminate bacteria on their skin and in their noses.”
The reported decision by county health officials not to inform the affected families of the hospital’s MRSA outbreak might be more sound and better argued (in my opinion) if the outbreak’s source (and the MRSA’s mode of transmission) had already been determined.
Once the source of a bacterial outbreak is known (and a root cause analysis performed), officials could then implement effective actions critical to the prevention of additional MRSA infections in the hospital’s NICU.
Whether it was reasonable for county health officials not to have informed the public and affected families sooner of this NICU’s potentially lethal MRSA outbreak, the source of which — for example, a colonized healthcare worker’s hands — still reportedly has not been identified, may certainly be debated.
Not the first time
The decision by health officials not to inform affected patients of an infection risk is not entirely uncommon.
By way of another example, at least one hospital in Los Angeles County has linked an outbreak of another type of organism called mycobacteria to a heater-cooler device.
While acknowledging the outbreak in February, however, officials of the Los Angeles County Department of Public Health have not warned potentially exposed surgery patients of the infection risk.
In fact, county health officials have declined to name the hospital where this mycobacterial outbreak occurred, The Los Angeles Times has reported.
The timely diagnosis and treatment of mycobacterial infections, as well as “close collaboration between device manufacturers and regulatory agencies to ensure safe use of the (heater-cooler devices), are essential to close this patient safety loophole,” researchers have emphasized.
Heater-cooler devices are used during certain open-chest surgeries to control and monitor the patient’s temperature, as required to perform the procedure successfully.
An in-depth discussion of these devices and the infection risk they can pose when contaminated is discussed in the related article, “Infections Are Not Limited to the LivaNova’s Sorin 3T Heater-Cooler Device or to Mycobacterium chimaera.”
Closing remarks
Dr. Susan S. Huang, who is one of UC Irvine’s physicians, co-authored an article in 2014 that provides strategies to prevent MRSA infections in hospitals (notwithstanding some of the disputed circumstances surrounding this same hospital’s MRSA outbreak affecting these 10 infants, as reported two weeks ago by the Los Angeles Times).
One of the strategies discussed in this 2014 article is the recommendation that patients and their families be educated about MRSA. According to this article, “Patient and family education should be provided as quickly as possible where the patient has a history of MRSA or once the patient’s MRSA-positive status has been detected.”
Whether UC Irvine strictly adhered to this recommendation beginning last summer in an effort to stop the spread of this MRSA outbreak in its NICU is unclear and in apparent dispute.
A related issue remains unresolved, understanding that MRSA’s mortality rate can be as high as 30%. According to hospital officials, all 10 of the infants infected (or colonized) with the outbreak’s MRSA strain have recovered.
But this assessment does not address whether one or more of the NICU’s infants might have tested positive for the outbreak’s MRSA strain and expired, but whose cause of death the hospital’s physicians attributed to a different factor, one that was unrelated to the outbreak’s bacteria.
This possibility could warrant further investigation, clarification and an independent review to determine whether any neonates who died since last August tested positive for MRSA.
Dr. Muscarella’s 5 most recently posted articles:
[catlist numberposts=5 author_posts=”muskiephd”]
Article by: Lawrence F Muscarella, PhD. Posted: April 25, 2017. LFM Healthcare Solutions, LLC Copyright 2017. LFM Healthcare Solutions, LLC. All rights reserved.
Lawrence F Muscarella PhD is the owner of LFM Healthcare Solutions, LLC, a Pennsylvania-based quality improvement and consulting company that provides safety services for hospitals, manufacturers and the public. Email Dr. Muscarella for more details.